Spinal cord injury is severe neurological condition in which the major mode of transmission between the brain and the body is disrupted. When higher levels of the spinal cord are injured, for example, in the neck, the injury can be immediately fatal. Those who survived spinal cord injury are often left paralyzed and at risk for a number of comorbid conditions such as pneumonia, depression, skin ulceration infection, urinary tract infections, and pain.
If patients who sustain spinal cord injury can receive medical treatment quickly, physicians may administer glucocorticoids to help reduce swelling around the injury and preserve spinal cord function. Patients may also undergo therapeutic hypothermia (a.k.a. targeted temperature management, whole body cooling), also to help reduce inflammation and prevent scar tissue from forming around the damaged spinal cord.
After the first few days to weeks after spinal cord injury, not much can be done to change the outcome of the disease. Patients may undergo intensive physical, occupational, and speech therapy to help regain function, but more often than not the neurological deficits are mostly permanent. Hence, researchers are feverishly searching for ways to treat spinal cord injury and, by extension, prevent or reduce paralysis and other chronic complications.
Mesenchymal stem cells are an intriguing potential therapy for spinal cord injury. These cells can easily be obtained from many different tissues including bone marrow and fat among others. In animals, mesenchymal stem cells have been shown to improve changes that occur during spinal cord injury, namely the regeneration and strengthening of nerve cells in the spinal cord. Research has also shown how adipose-derived stem cells are a potential option for those with neurological conditions such as spinal cord injury.
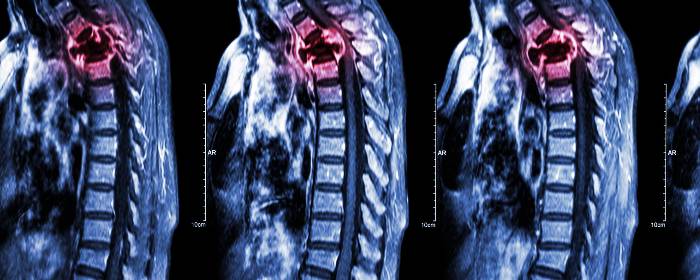
To test this possible effect in humans, researchers collected mesenchymal stromal (stem) cells from patients with spinal cord injury in their upper back (i.e. thoracic spinal cord). Researchers then prepared and administered those cells back into the cerebrospinal fluid of the same patients. Each patient received two or three injections of approximately 1,000,000 cells per kilogram body weight. There were no adverse effects of the treatment for up to two years after injection. MRI imaging showed no abnormalities resulting from stem cell infusion. While the authors write that there were too few patients to make any firm conclusions about the efficacy of the treatment, they were strongly encouraged by the safety of the procedure. In fact, they use these results to begin a placebo-controlled clinical trial.
Reference
Satti et al. (2016). Autologous mesenchymal stromal cell transplantation for spinal cord injury: A Phase I pilot study. International Society for Cellular Therapy, 18(4),518-522.

 St. Petersburg, Florida
St. Petersburg, Florida
